Note: I said Stupendous not Stupid.
What is a science fair project? A Model, a collection or an experiment are the three types of projects.
A model is ideal for an elementary student. In all fairness, models are used by scientists to represent objects too small, too large, too expensive or unobtainable to work with. Watson and Crick cracked the molecular structure of DNA with a model. Models are not to be dismissed. However, the average model made by students do not use what is called higher order thinking. This is why it is still suitable for an elementary student but not a high school student.
 |
| Volcano erupting on Montserrat |
Models made may be
1. plant or animal cells
2. isotopes or atom structure
3. solar system
4. mature versus young river systems
5. the ever popular "Volcano"
Scientifically, a model as well as careful observation and description of objects is very relevant.
Being able to note slight variations that enable classification is the hallmark of creating collections and organizing information.
Examples of collections would be
1. leaves
2. insects
3. sea shells
4. rocks
An experiment is usually requested as a science project because it demonstrates an understanding of the Scientific Method.
The Scientific Method is no small concept in that it revolutionized the world and how it processed information. The scientific method is essentially the reason why we know the Sun and not the Earth is the center of the solar system, many diseases are caused by germs, traits of offspring are inherited from parents and horsehair does not turn into worms when it rains. The scientific method essentially says, prove it to me and prove it to me over and over. The scientific method can be summed up in seven steps.
Seven Steps of the Scientific Method.
STEP 1: STATE YOUR PROBLEM
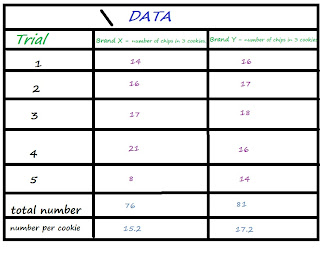
Sample Experiment: Which brand of Chocolate Chip Cookie has the most chocolate chips?
This is actually the hardest part of a science fair project. Developing an experiment. As a judge, a learner, an observer, how interesting is a project in which you know the outcome.
The following is one of the most overdone projects. It entails the purchase of two plants. One plant is treated normal and the other plant is either deprived of light or water or poisoned with salt or sugar. What makes this experiment such a poor choice is two fold.
1. Who cares. Everyone knows the answer. The creator really does not care about their work, they just want to get it done.
2. Two plants are not enough to prove or disprove anything. You need to repeat an experiment several times and have several plants to remove the possibility of a fluke or coincidence.
Take your time. Think about what you are really interested in. When I went to a state Science Fair in Georgia, the rural students had very good projects. The students I accompanied where from a suburb of Atlanta and they were surprised such small town kids did so well. What the rural students did was have very practical observations. For example, one students explored why algae did not grow on a tin roof. One of the students I accompanied did an experiment where they tested how plants grew without gravity. Both were good projects to be there. The one that related to everyday concerns won.
Another consideration, how many people are doing the same project. If 12 students do the same project, their project does not stand out as well. You better make the project incredibly attractive to make a judge want to pick your project. You also have to do every step exceedingly well.
Now you have read and read and you still can't think of an experiment. Pick a subject area you are interested in. Perhaps you will have a question yourself. A big part of creativity is giving yourself time to think. Many people think, I'm not creative. Well creativity is not done like a quiz game at school where speed of answer is at issue. Creativity is giving yourself time to think. Mow grass, do chores, it will come to you.
Depending on your age, you might use a book of proposed experiments. Once you get to high school, you need to be thinking in terms of what would be a great experiment. It is still OK to choose your experiment from a resource. As you study science you will find areas you want to explore more.
You could build your own computer which would be a model. You could compare brands of ports or processor chips which changes your project into an experiment.. The big drawback to this experiment would be the expense. You could take discarded computers to rebuild a computer. You could take computers that are beyond salvage and compare their power supply or motherboard with their computing capacity.
Cost is an issue. Real scientist take into account how much an experiment will cost. The student who did the anti-gravity experiment ran the family electric bill up $200 more the month he did the experiment. If a science project is good, it will be good regardless of how little it cost to do.
It's the process that you are demonstrating. When I went to college, I was so thrilled. No homework was assigned. Then the real discovery was I found myself creating homework so I could do well on the tests. When I was stumped, I had to go to the professor before or after class or sometimes during class if they took questions. The same is true with scientific discovery. The ability will come.
Last but not least, State your problem in the form of a question.
STEP 2: RESEARCH THE PROBLEM
While researching the problem which brand of chocolate chip cookies has the most chips, I browse the cookie aisle of the grocery store. I looked at the size of the cookie on the package, I count the chips on the cookie shown, I look at the calories per cookie, I estimate the weight of each cookie, I look at the fat grams. Fat grams you say. Of course, chocolate is cocoa butter, cocoa and sugar. The most fat, possibly the more chips. That type of knowledge can come from the library or say your mom. Discussing the problem with knowledgeable people can give you clues on how to approach your problem.
Researching your problem also indicates whether your problem is a good one to tackle or perhaps you should think of another problem. Look at whether you can obtain the supplies needed to do the experiment. Examine the cost. Do you have a good area to do the project. If you do the project in the dead of winter, you cannot choose a project which only works when it is 90 degrees Fahrenheit or 32 degrees Celsius outside. Real scientist are practical.
Why would you choose packaged goods versus Aunt Sue's or Cousin Meggie's to compare. Aunt Sue and Cousin Meggie are not going to make enough cookies to satisfy whoever has a whim for their cookies. Plus they probably vary their recipe depending on what is on hand. Your experiment needs to be duplicated by other scientists.
STEP 3: FORM AN HYPOTHESIS
Make a prediction as to what the answer to your question will be. An hypothesis is a statement that can be tested not an opinion.
Write down the answer to the question you originally posed. This is called your hypothesis. An hypothesis is defined as a scientific guess, educated guess or most recently a testable explanation for a problem.
My hypothesis is that brand X will have more chocolate chips per cookie than brand Y. I decided this because brand X has more fat grams and the picture on the package has 23 visible chips and brand Z has 12 visible chips.
STEP 4: DESIGN AN EXPERIMENT TO TEST YOUR HYPOTHESIS
When creating an experiment, you are comparing two factors that are identical except one factor has been changed. The factor you change is called the variable. It is also called your independent variable. All conditions between the two trials must be identical or as close as possible. You keep notes as to what could not be kept the same. The variables that do not change are noted and called controlled variables.
Perhaps the experiement of "Which brand of chocolate chip cookie has the most chips?" has controlled variables as being done at the same temperature, the same scientist, the same speed of experimentation, the same light level.
The variable that may differ are the size of the cookie or size of the chip.
The experiment may be conducted by taking a cookie from each brand and breaking up the cookie and counting the amount of chocolate chips. You need some sort of measurement or observation not an opinion. An opinion would be it seemed to shake. You need something quantifiable which means measured. It vibrated 2 cm in each direction for 20 seconds would be a measurement.
Stating that one object is prettier than another is a value judgement and cannot be measured. However description of a smell as unpleasant or pleasant would be a valid observation.
STEP 5: COLLECT DATA
A chart or table is preferable. A great deal of information can be stored in a logical easy to find format.
Scientist go for the practical and expedient. A great riff of a guitar is art. When you write excessively, it is difficult to read. Even poets, novelist, journalist write only what is necessary to tell their story. Anything extra distracts from what is told.
This is the fun part of an experiment. You may have to conduct your experiment over a period of several days. What you will have to do is conduct the experiment several times. In order for results to be valid, you must get similar results over and over.
STEP 6: FORM A CONCLUSION
State whether the hypothesis was proven or proved wrong.
In addition, you should discuss factors that affected your experiment. How could these have affected the results. This is good area to propose variations of the experiment to get better results.
STEP 7: PUBLISH
Essentially this is what your science project display is all about. Your teacher will give specific guidelines for the science fair.
Remember, keep it attractive. Attractive projects score better than the science behind them. Go into a store, products are placed in packages designed to make you want them. Impulse items are on corners or eye level. Why do you think they put candy, gum, soft drinks, nuts and magazines at the checkout counter of a grocery.